Now, the Guidelines International Network (G-I-N) has launched a large-scale toolkit aimed to address commonly experienced issues related to patient and public involvement. The result of a combination of international experiences and best practice examples, the toolkit is a one-stop shop spanning the systematic review and guideline development process, from conducting targeted consultation with the public to recruiting and supporting patient panel members to communicating recommendations to the public at-large. As a "living resource," the toolkit will continue to expand and evolve as further information and experience is cultivated.
Friday, January 7, 2022
Guideline Development Resource Alert: the G-I-N Public and Patient Toolkit
Now, the Guidelines International Network (G-I-N) has launched a large-scale toolkit aimed to address commonly experienced issues related to patient and public involvement. The result of a combination of international experiences and best practice examples, the toolkit is a one-stop shop spanning the systematic review and guideline development process, from conducting targeted consultation with the public to recruiting and supporting patient panel members to communicating recommendations to the public at-large. As a "living resource," the toolkit will continue to expand and evolve as further information and experience is cultivated.
Monday, December 20, 2021
2021 Reflections Before a Big Year Ahead
From all of us at the U.S. GRADE Network, thanks for making 2021 a year to remember.
- We hosted our 14th and 15th workshops virtually - meaning we had the joy of talking GRADE with 95 attendees from many corners of the world, including Spain, Argentina, Pakistan, and Peru.
- We co-published an international needs assessment of guideline-producing organizations in collaboration with the Guidelines International Network.
- We announced our first ever two-day comprehensive systematic review workshop scheduled to be held virtually this coming February 1-2. Learn more and register here.
- And we continue to look forward to hosting our 16th guideline development workshop, once again live and in person, from June 29-July 1, 2022, in Chicago, Illinois. Registration now open.
 |
| GRADE faculty take a moment to pose for a picture with some attendees of the fall 2021 virtual guideline development workshop. |
Thursday, November 11, 2021
Fall Scholars Dazzle at the 15th GRADE Guideline Development Workshop
During the 15th GRADE Guideline Development Workshop held virtually last month, the Evidence Foundation had the pleasure of welcoming three new scholars with the opportunity to attend the workshop free of charge. As part of the scholarship, each recipient presented to the workshop attendees about their current or proposed project related to evidence-based medicine and reducing bias in healthcare.
Razan Mansour, MD, a postdoctoral research fellow at the University of Kansas Medical Center, spoke about the challenges of navigating variabilities among published systematic reviews when developing clinical recommendations. Variability may emerge from differences between reviews regarding study inclusion criteria, risk of bias assessment, and the way data are presented. Most sources of variability, Dr. Mansour said, are difficult to explain, but a modified version of the A MeaSurement Tool to Assess systematic Reviews (AMSTAR) 2 tool can help identify common "red flags" and help to prioritize and identify the best systematic reviews off of which to base resulting clinical recommendations. Information from multiple higher-quality systematic reviews may be used, and data from individual studies may need to be extracted to paint a complete picture of the evidence.
Next, Reena Ragala, MOTR/L, spoke about her project as a Senior Evidence-Based Practice Analyst at the Medical University of South Carolina Value Institute. The Institute was established in 2012 to support MUSC's efforts to build the infrastructure to advance science and discovery through evidence-based practice. Starting in the spring/summer of 2022, Ragala will help lead guideline development "boot camps" though the Institute to train providers within rural health networks in the application of evidence-based guideline methodology. Through this process, Ragala hopes to empower rather than intimidate front-line staff and clinicians new to clinical guideline development.
"When using GRADE for diagnostic tests or guideline development, judgements or recommendations are based on patient outcomes, said Ragala. "GRADE doesn’t just look at the strength/quality of the evidence, but incorporates the feasibility, accuracy, bias, and benefit/harm to the patient to ultimately make recommendations."
Carolina Soledad, MD, MStat, PhD, spoke about the unique opportunities, challenges, and solutions within an aspect of guideline development that has gained great relevance in recent years: reaching consensus within the context of virtual meetings. Dr. Soledad, a junior methodologist within the European Society of Anesthesia and Intensive Care (ESAIC) Guidelines Committee, discussed the fact that while in-person meetings have several strengths - such as access to nonverbal cues such as body language and facial expression and shared context - virtual meetings also have unique benefits, such as cost savings and a reduced need to focus on logistics. In addition, they have become a necessity in the age of a global pandemic. To help address the unique questions surrounding virtual meetings - such as the optimal length and number of participants - Dr. Soledad developed a 33-item survey of anesthesiologists and intensivists involved in guideline development. The findings will help formulate best practices for improving the level of communication, engagement, and effectiveness of future virtual meetings.
According to Dr. Soledad, "This workshop gave me a peek into GRADEpro GDT software, and it turned out to be easier to use than I've thought!"
If you are interested in learning more about GRADE and attending the workshop as a scholarship recipient, applications for our upcoming workshop in Chicago, Illinois, are now open. The deadline to apply is March 31, 2022. Details can be found here.
Friday, September 24, 2021
6 Simple Rules for Creating a Plain Language Summary of a GRADE Guideline Recommendation
While seasoned clinicians and methodheads may consider the nuances of clinical guideline development everyday fare, there is generally a lack of awareness among the public about the implications and use of guidelines. In addition, there is some evidence of public concern that guidelines may be used to ration care as well as public confusion about how they should be applied to an individual's unique needs. The translation of guideline recommendations into plain language, however, may help improve public knowledge and awareness of the applications and implications of guidelines.
In a new paper published in the Journal of Clinical Epidemiology, Santesso and colleagues set out to develop a template for communicating guideline recommendations as well as explore public attitude around guidelines. First, the authors conducted semi-structured focus groups to gather information about general perceptions and opinions regarding guidelines. Then, these insights were used to develop a plain language template which was user-tested. The template was then revised into a final version. During the process, a few key themes emerged, including:
- an upfront and clear description of the population/individuals to whom the guideline applies
- a section detailing topics and questions to bring up with one's health care provider
- definitions surrounding the strength of the recommendation, and further considerations for decision-making around conditional recommendations
- formatting that makes use of bullets and tables rather than blocks of text
- the recommendation, its strength (with a symbol), and an explanation
- the population/individuals to whom the recommendation applies
- rationale for the strength of the recommendation
- additional considerations when using the recommendation
- benefits and harms
- implications, what a patient can do, and questions or topics to discuss with one's health care provider
Monday, September 13, 2021
Re-analysis of a systematic review on injury prevention demonstrates that methods do really matter
How much of a difference can methodological decisions make? Quite a bit, argues a new paper published in the Journal of Clinical Epidemiology. A re-analysis of a 2018 meta-analysis on the role of the Nordic hamstring curl (NHE) on injury prevention, the study outlined and then executed several methodological changes within the context of an updated search and found that the resulting magnitude of effect - and strength of recommendations using GRADE - were not quite as dazzling as the original analysis.
Impellizzeri and colleagues noted several suggested changes to the 2018 paper, including:
- limiting the meta-analysis to higher-level evidence (randomized controlled trials) when available,
- clarifying the interventions used in the included studies and being cognizant of the effect of co-interventions (for instance, when NHE was used alone versus in combination with other exercises as part of an injury reduction program),
- being careful not to "double-dip" on events (i.e., injuries) that recur in the same individual when presenting the data as a risk ratio
- discussing the impact of between-study heterogeneity when discussing the certainty of resulting estimates,
- presenting the lower- and upper-bounds of 95% confidence intervals for estimates of effect in addition to the point estimates, and
- taking the limitations of the literature and other important considerations into account when formulating final summaries or recommendations (for instance, using the GRADE framework)
Monday, August 30, 2021
Misuse of ROBINS-I Tool May Underestimate Risk of Bias in Non-Randomized Studies
Although it is currently the only tool recommended by the Cochrane Handbook for assessing risk of bias in non-randomized studies of interventions, the Risk Of Bias In Non-randomized Studies of Interventions (ROBINS-I) tool can be complex and difficult to use effectively for reviewers lacking specific training or expertise in its application. Previous posts have summarized research examining the reliability of ROBINS-I, suggesting that it can improve with training of reviewers. Now, a study from Igelström and colleagues finds that the tool is commonly modified or used incorrectly, potentially affecting the certainty of evidence or strength of recommendations resulting from synthesis of these studies.
The authors reviewed 124 systematic reviews published across two months in 2020, using A MeaSurement Tool to Assess systematic Reviews (AMSTAR) to operationalize the overall quality of the reviews. The authors extracted data related to the use of ROBINS-I to assess risk of bias across studies and/or outcomes as well as the number of studies included, whether meta-analysis was performed, and whether any funding sources were declared. They then assessed whether the application of ROBIN-I was predicted by the review's overall methodological quality (as measured by AMSTAR), the performance of risk of bias assessment in duplicate, the presence of industry funding, or the inclusion of randomized controlled trials in the review.
Overall methodological quality across the reviews was generally low to very low, with only 17% scoring as moderate quality and 6% scoring as high quality. Only six (5%) of the reviews reported explicit justifications for risk of bias judgments both across and within domains. Modification of ROBINS-I was common, with 20% of reviews modifying the rating scale, and six either not reporting across all seven domains or adding an eight domain. In 19% of reviews, studies rated as having a "critical" risk of bias were included in the narrative or quantitative synthesis, against guidance for the use of the tool.
Reviews that were of higher quality as assessed by AMSTAR tended to contain fewer "low" or "moderate" risk of bias ratings and more judgments of "critical" risk of bias. Thus, the authors argue, incorrect or modified use of ROBINS-I may risk underestimating the potential risk of bias among included studies, potentially affecting the resulting conclusions or recommendations. Associations between the use of ROBINS-I and the other potential predictors, however, were less conclusive.
Igelström, E., Campbell, M., Craig, P., and Katikireddi, S.V. (2021). Cochrane's risk-of-bias tool for non-randomized studies (ROBINS-I) is frequently misapplied: A methodological systematic review. J Clin Epidmiol, in-press.
Manuscript available from publisher's website here.
Tuesday, August 24, 2021
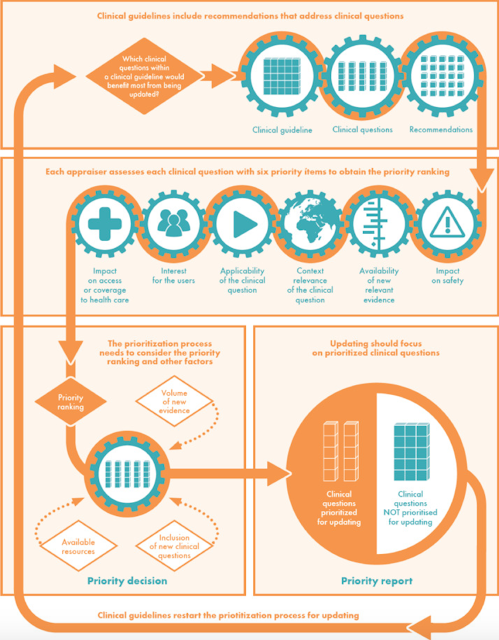
UpPriority: A new tool to guide the prioritization of guideline update efforts
- the potential impact of an outdated guideline on patient safety;
- the availability of new, relevant evidence;
- the context relevance of the clinical question at hand (is the question still relevant given considerations such as the burden of disease, variation in practice, or emerging care options?);
- methodological applicability of the clinical question (does the question still address PICO components of interest?);
- user interest in an update; and
- the potential impact of an update on access to health care.